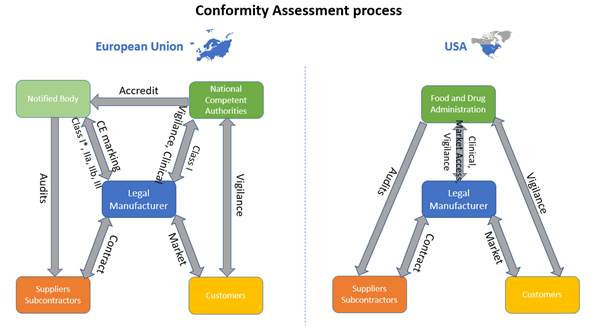
What are the principal differences between the conformity assessment process of a medical device in the USA and in the European Union? - Kvalito

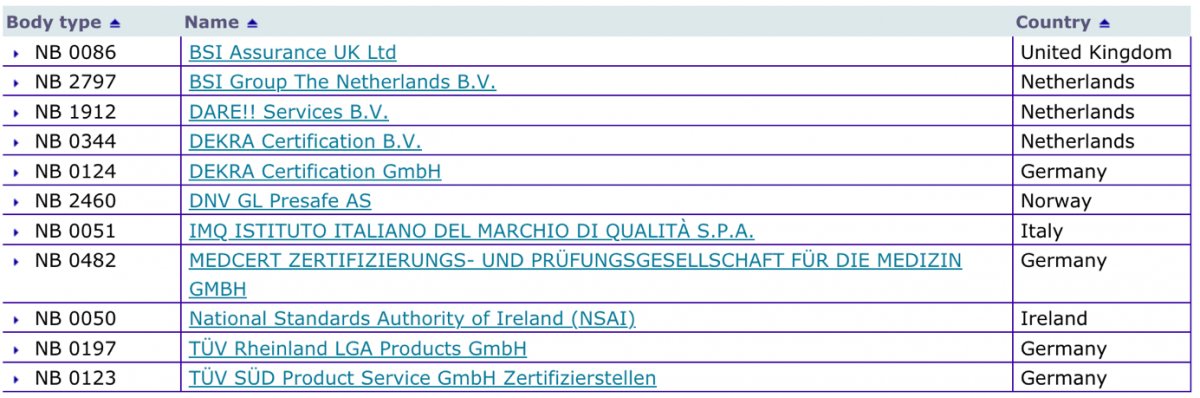
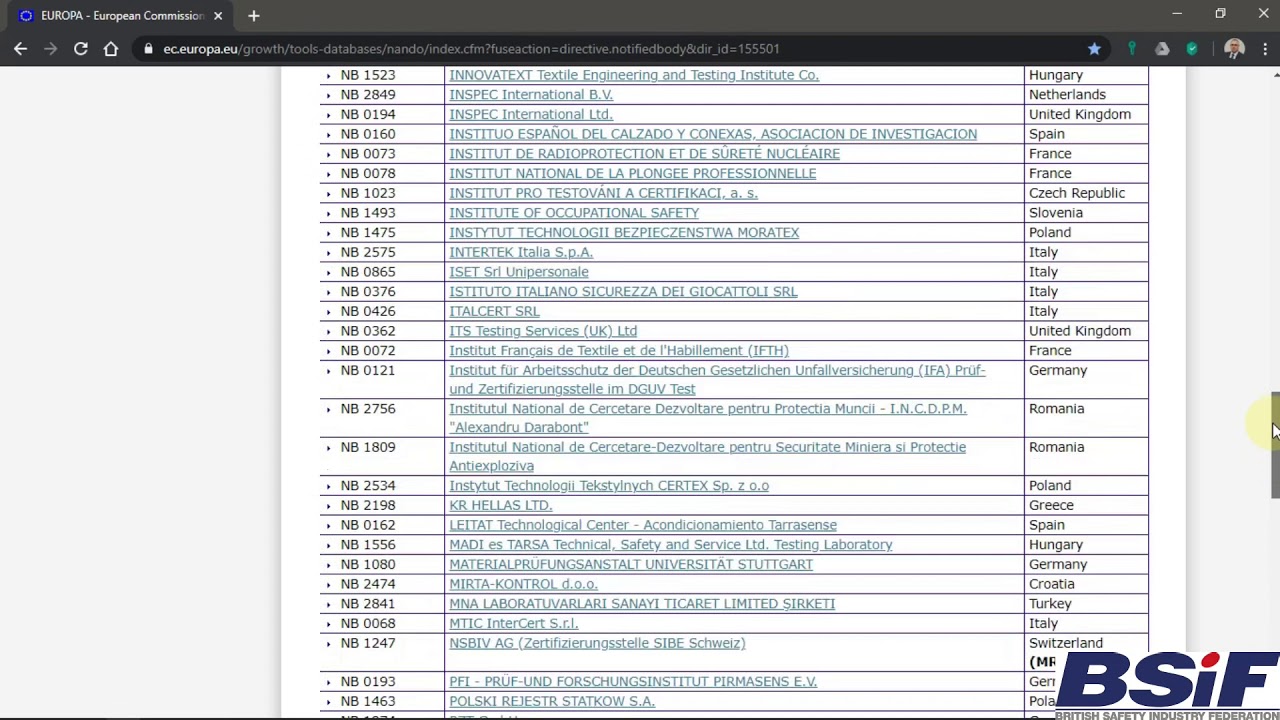
Explosives Working Group 4 November 2014 Notified Bodies and NANDO Norma McGovern, DG ENTR.C1 – Internal Market and its International Dimension. - ppt download